 
This review will underline the advantages of core–shell silica particles in the analysis of pharmaceutically active ingredients based on liquid chromatography from the perspective of column properties, system suitability test parameter results and validation steps.Background. 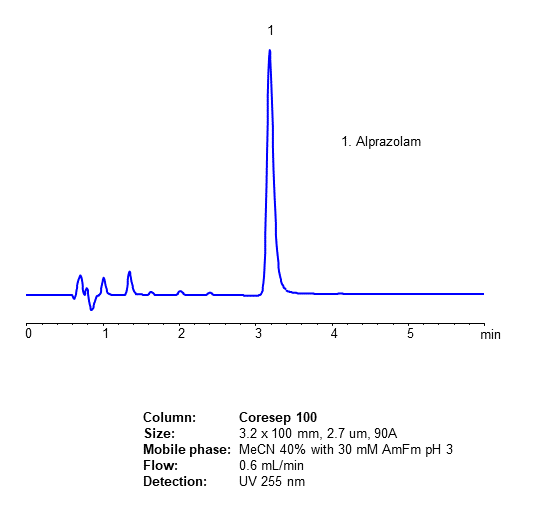
The columns packed with core–shell particles have been employed in a wide range of applications for analysis and quality control of pharmaceutical active substances. The key factors for core–shell particles are size and thickness of porous shell layer, the latter of which can be explainĮd using the Van Deemter equation. Thus, core–shell technology provides the same efficient separations as the sub 2 µm particles that are used in UHPLC, while eliminating the disadvantages (potentially lower backpressure). However, in recent years, core–shell silica particles (a combination of solid core and porous shell) have been increasingly used for highly efficient separation with reduced run times. Stationary phases that have fully porous silica particles comply with the essential criteria of analysis, but these show all the limitations of HPLC. In general, two different silica types of column packing material based on their backbone have been used for HPLC and UHPLC. To overcome these limitations, scientists have developed new type of column particles. On the other hand, they have some limitations: In some cases, traditional HPLC uses high amounts of organic solvents with longer analysis time, and furthermore UHPLC has high back pressure and frictional heating. Both techniques are preferred due to their selectivity, high accuracy and remarkable precision. The most important challenge in these techniques is fast and efficient separation. High performance liquid chromatography (HPLC) and ultrahigh performance liquid chromatography (UHPLC or UPLC) have been the most widely used tools for research and routine quality control of active pharmaceutical ingredients (API). This review will underline the advantages of core–shell silica particles in the analysis of pharmaceutically active ingredients based on liquid chromatography from the perspective of column properties, system suitability test parameter results and validation steps. The key factors for core–shell particles are size and thickness of porous shell layer, the latter of which can be explained using the Van Deemter equation.  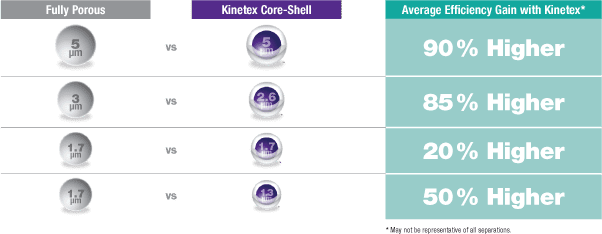 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
